Ithaca, New York:
Behaviour instead of Identity: Functional Complexity of Organic Matter as an Organising Principle in Soil Ecosystems
Essay
Organic matter in soil possesses an enduring allure. This is not only because soil houses the largest pool of carbon in terrestrial ecosystems – more than all plants on earth and the global atmosphere together. Nor is it solely because organic matter is the most important indicator of soil’s health. Organic matter is also alluring because it escapes easy characterisation. After centuries of scientific investigation, the research community is still arguing about what comprises organic matter in soil and why it exists in the first place. The prevailing no- tion (even though specialised textbooks begin to change their treatment of the subject) is that soil organic matter consists of humus composed of various forms of humic substances and that the identity of these humic substances determines a particular soil’s behaviour. Here I critique the idea of ‘humus’ and the focus on identity that goes along with that idea in favour of a focus on behaviour. Such a shift in perspective allows fundamental questions to be asked about the role of functional complexity between (rather than the identity of ) constituents in soils as well as interactions in ecosystems in general, including in the human societies.
In this essay I argue that our questions also hold on to outdated notions of soil: when we ask ‘how can we stop losing organic matter?’ or ‘how can we add more organic carbon to sequester atmospheric carbon dioxide?’ we are mobilising an idea of soil as being controlled by a specific chemical identity (i.e., humus) that does not correspond to how soil actually works. I posit that the behaviour of soil organic matter determines its identity, not the other way round.
A leaf added to a Petri dish mixed with soil microorganisms in the lab de- composes remarkably quickly: within days or weeks, the organic carbon in that leaf is fully turned into carbon dioxide. In contrast, the same organic carbon atom from that leaf added to an actual plot of soil may remain detectable in that soil as an organic compound for millennia. What explains this dramatic difference? The historical explanation was ‘humification’. Humification was the process by which microorganisms in soil eat (a scientist would say ‘metabolise’) small bits and pieces of decomposing plant residues to make large and complex molecules (containing many different chemical bonds). In older textbooks these newly-made molecules are called ‘humic substances’. And scientists surmised that these humic substances (in some theories, together with ‘non-humic sub- stances’) comprise ‘humus’. Such ‘humic substances’ accrue over time in soil because they are recalcitrant in both their size and chemical composition: too big or too ‘complex’ in their identity to be eaten by microorganisms.
Despite its popularity, there were always conceptual and analytical challenges to the humic paradigm: why would microorganisms invest energy in making these large and complex molecules? It made no ecological sense. Secondly, the proof that humic substances existed and their quantification and isolation rested solely on the fact that they were soluble and extractable in an alkaline solution (Kleber and Lehmann 2019) An extraction is a process whereby a certain proportion of matter is brought into solution for further analysis. Think of your tea or coffee, which are extracts of tea leaves or coffee beans. Not all the tea leaves or beans are in the solution, just whatever is soluble in hot water that we drink. Some of the tea leaves or coffee grounds remain in the tea bag or filter paper. This is in some ways selective (only what is soluble in hot water) and at the same time general (a lot of different molecules in a tea leaf are soluble in hot water). Extraction on its own may therefore not be a suitable approach for defining a substance (as done using an alkaline extraction to define ‘humic substances’).
The history of humic substances research is long and not in all its facets sufficiently explored. It is generally assumed that Carl Friedrich Achard is the scientist who came up with the approach of using alkali solution to extract organic matter from soil in the late eighteenth century (Achard 1786). Achard was actually only interested in examining the value of peat as a fuel, and did not use the method he came up with to define humic substances. That came decades later and we do not need to go into further detail here about how the method of extraction became the definition of humic substances.
With the onset of modern spectroscopy and spectromicroscopy, which allowed us to look more closely into soil, it became clear that we did not need the notion of humic substances to explain soil organic matter. It was sufficient to consider known decomposition products from soil biota. With the tools of spectroscopy and microscopy, we could observe that soil is a complex mixture of compounds that microorganisms make (their own biomass, exudates, enzymes, etc.). We could now understand the reason for their production as a result of a living microbial population, of the need to make enzymes to access nutrients and carbon or to get rid of toxic material, not as a new substance for which the ecological reason remains unexplained. Furthermore, ‘humic substances’ as defined by extraction using alkali solution do not exist at the molecular scale at which they should occur (Lehmann et al. 2008). When we looked for evidence of the chemical signature of what can be extracted by an alkali solution (and what is then called ‘humic substance’) with an electron microscope, we came up empty-handed.
Why is it important to know whether ‘humic substances’ exist or not? If the recalcitrance of a substance (as in, its ability to resist being eaten by microorganisms because of its chemical structure, as described for ‘humic substances’) is not the reason for the accumulation of organic carbon in soil, what is? Finding the answer to this question helps us also understand how to better manage and care for our soils.
Given contemporary biogeochemical evidence, we should rather think of soil as a spatially and compositionally highly complex ecosystem that is constantly in flux, where temperature and moisture change all the time and often change rapidly. Organic carbon can accrue because it is not metabolised to carbon dioxide, and not because it cannot be metabolised (Lehmann et al. 2020). Such a shift in thinking would mean that we cannot truly ‘sequester’ or ‘lock up’ carbon in soil, akin to using a key that locks a door to store our carbon, and walk away – but have to instead adopt a notion of ‘constant care’ (Janzen 2024), whereby we curate the inputs and outputs from the soil. This is what Henry Janzen (2024) calls ‘stewardship’. By adding more carbon and losing less, we maintain or possibly increase the organic matter in soil that is truly a flow-through reactor of transient carbon rather than a storage container of static carbon (Figure 1).
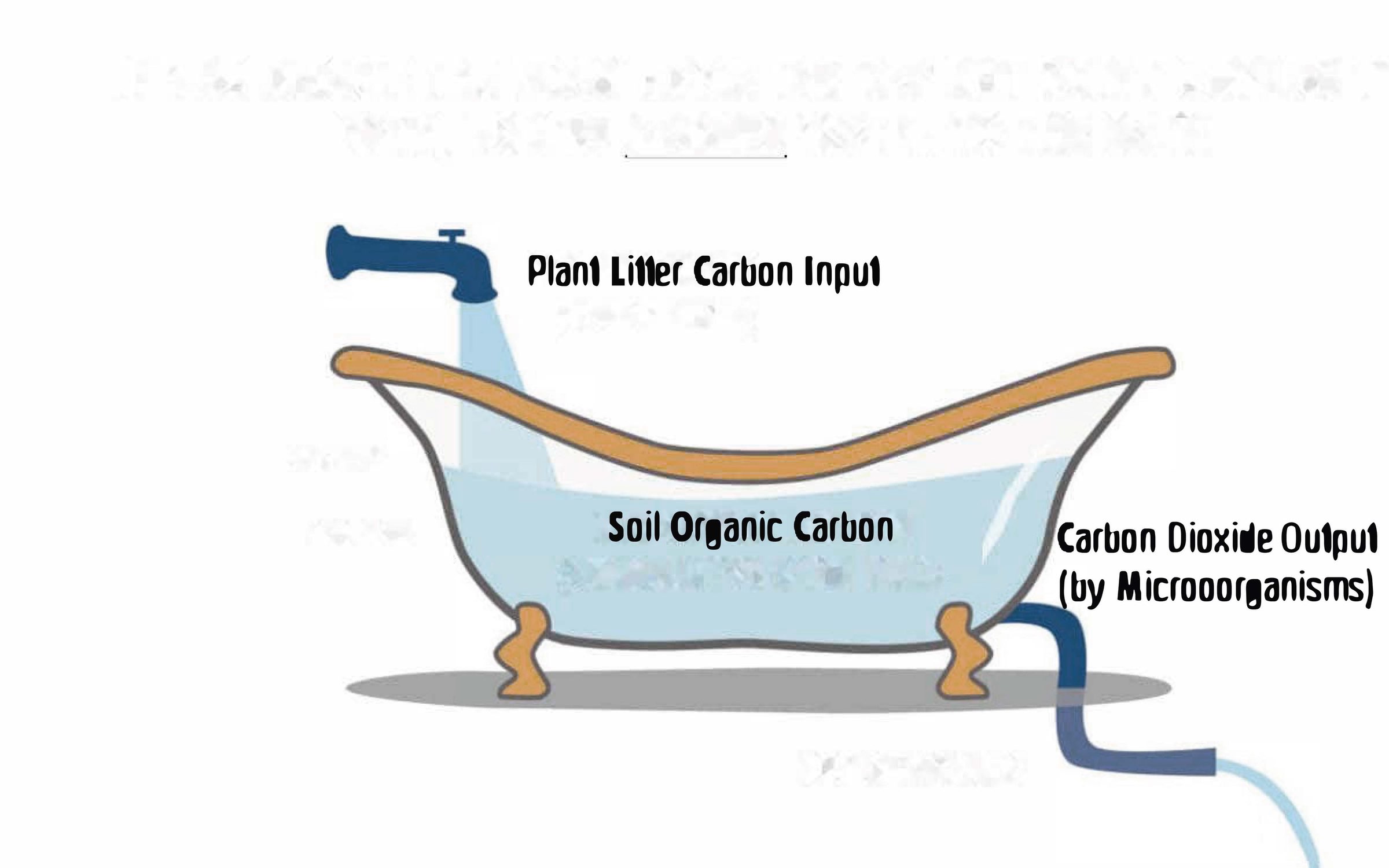
Figure 1. Bathtub analogy of why there is soil organic carbon as a balance between input (plant input of photosynthetically fixed carbon) and output (carbon dioxide). Soil organic carbon can flow out of the bathtub in principle, but it does not, because the pipe going out is controlling the output.
Another important aspect that must be reassessed when questioning extracted ‘humic substances’ is that an ‘average soil’ does not exist. Using very fine-scale investigation with the help of synchrotron radiation (in essence a very powerful electron microscope that not only gives us the spatial features of soil at 10s of nanometres, way finer than a human hair, but also a lot of information about its chemical composition) it became clear that the average composition of soil organic matter cannot be found at a fine spatial scale, where these substances should occur. This is a profound and important insight with far-ranging implications for our understanding of why soil organic matter exists and how we can manage soils for crop production as well as other soil health aspects including carbon accrual and consequent climate change mitigation.
We can visualise this with a simple experiment of making a fruit smoothie: take three different fruits (apple, pear, melon?) and mix them together in a blender (Figure 2). The first question is: have we made a new fruit? No. The smoothie is still a mixture of three fruits, not a new fruit. When we drink the smoothie, we sense a taste that is not only very difficult to identify (try it with your friends), but also has a taste distinct from the three fruits individually – even though we have made no new fruit. We made a taste that in some ways is an average of the three fruits (not exactly, but that is not important for the argument being illustrated here). At the same time, we know that this taste is a mixture of existing tastes, not new fruit. And if someone is allergic to apples, it is important to know that there are still apples in the smoothie. In the same way in soil biogeochemistry, we have to know what compounds are in the complex mixture of soil organic matter, and the average composition is not sufficient to explain interactions between actually existing molecules and minerals or microorganisms, because the average fruit does not exist in a smoothie. Similarly, the average in this situation is a meaningless way of looking at and explaining soil organic carbon properties and behaviour, and ‘humic substances’ extracts will always be an average of what can be extracted. Since there is no other method to characterise, identify or quantify ‘humic substances’ than by an alkaline extraction, ‘humic substances’ are not a helpful way to truly understand soil organic carbon.

Figure 2. Fruit smoothie experiment as a performance on ‘humic substances’ at the workshop ‘Thinking from the Substrate’, 3–6 June 2024, Harnack House, Max Planck Institute, Berlin, organised by Emily Brownell. Photos: courtesy of Amiel Bize (left); by the author (right).
The shift in scholarship from sequestering recalcitrant ‘humic substances’ to caring about feeding and housing diverse actors with diverse substrates is much more in line with Donna Haraway’s assertion that ‘Human as humus has potential’ (Haraway 2016). I read Haraway’s essay as a proposal to see humans as a part of an ever-changing ecosystem with numerous interactions between a diverse set of actors, analogous to soil with its myriads of different microbes. In the new scientific view beyond ‘humus’, the persistence and resilience of soil organic matter is a product of what we called ‘functional complexity’ (Lehmann et al. 2020). This means persistence no longer has to do with material recalcitrance as a result of size (e.g., too big to be eaten by microorganisms); and it also means that there is not a one-time fix with which we can ‘lock carbon away’.
This shift to a view consistent with care rather than size or ‘strength’ not only aligns better with experimental evidence but is also more in line with historical and contemporary attitudes towards soil and soil health. I therefore propose to see organic matter persistence as a question about behaviour rather than identity. What I mean by ‘behaviour’ includes mineral interaction (such as adsorption) or solubility, as well as the movements of molecules or soil biota that are all known to be pivotal in determining how much organic matter is in soil. By contrast, the ‘identity’ of soil refers to its molecular size or chemical bonds, or its elemental composition. Behaviour is influenced by identity, but not solely determined by it: the extent of adsorption of a molecule to a clay particle will be strongly influenced by whether the molecule is positively or negatively charged or not charged at all, but also clay properties will matter, such as the amount of their surface area, and whether the soil is dry or wet, cold or warm, all environmental properties (Schmidt et al. 2011). What the persistence of soil organic matter is, will then be a result of many factors: it is therefore not its property or identity but its behaviour that we need to monitor and curate.
A shift in scholarship is therefore warranted for many reasons. And this proposed shift (Lehmann and Kleber 2015) would not only be based on observational evidence and logic but also allow a more meaningful and forward-looking engagement with soil processes as argued in this essay. Would such a shift also change how we view human identity and behaviour?
References
- Achard, F.C. 1786. ‘Chemische Untersuchung des Torfs’. Crell’s Chemical Annals 2: 391–403.
- Haraway, D. 2016. ‘Tentacular Thinking: Anthropocene, Capitalocene, Chthulucene’. E-Flux Journal 75: 8/16.
- Janzen, H.H. 2024. ‘Soil Carbon Stewardship: Thinking in Circles’. European Journal of Soil Science 75: e13536.
- Kleber, M. and J. Lehmann. 2019. ‘Humic Substances Extracted by Alkali Are Invalid Proxies for the Dynamics and Functions of Organic Matter in Terrestrial and Aquatic Ecosystems’. Journal of Environmental Quality 48: 207–16.
- Lehmann, J., D. Solomon, J. Kinyangi, L. Dathe, S. Wirick and C. Jacobsen. 2008 ‘Spatial Complexity of Soil Organic Matter Forms at Nanometre Scales’. Nature Geoscience 1: 238–42.
- Lehmann, J. and M. Kleber. 2015. ‘The Contentious Nature of Soil Organic Matter’. Nature 528: 60–68.
- Lehmann, J., C.M. Hansel, C. Kaiser, M. Kleber, K. Maher, S. Manzoni, N. Nunan, M. Reichstein, J.P. Schimel, M.S. Torn, W.R.Wieder and I. Kögel-Knabner. 2020. ‘Persistence of Soil Organic Carbon Caused by Functional Complexity’. Nature Geoscience 13: 529–34.
- Schmidt, M.W.I., M.S. Torn, S. Abiven, T. Dittmar, G. Guggenberger, I.A. Janssens, M. Kleber, I. Kögel-Knabner, J. Lehmann, D.A.C. Manning, P. Nannipieri, D.P. Rasse, S. Weiner and S.E. Trumbore. 2011. ‘Persistence of Soil Organic Matter as an Ecosystem Property’. Nature 478: 49–56.
